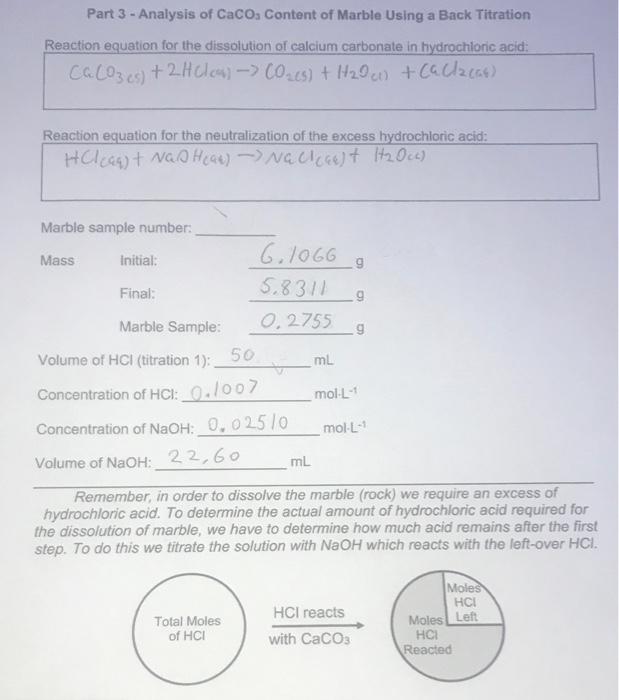
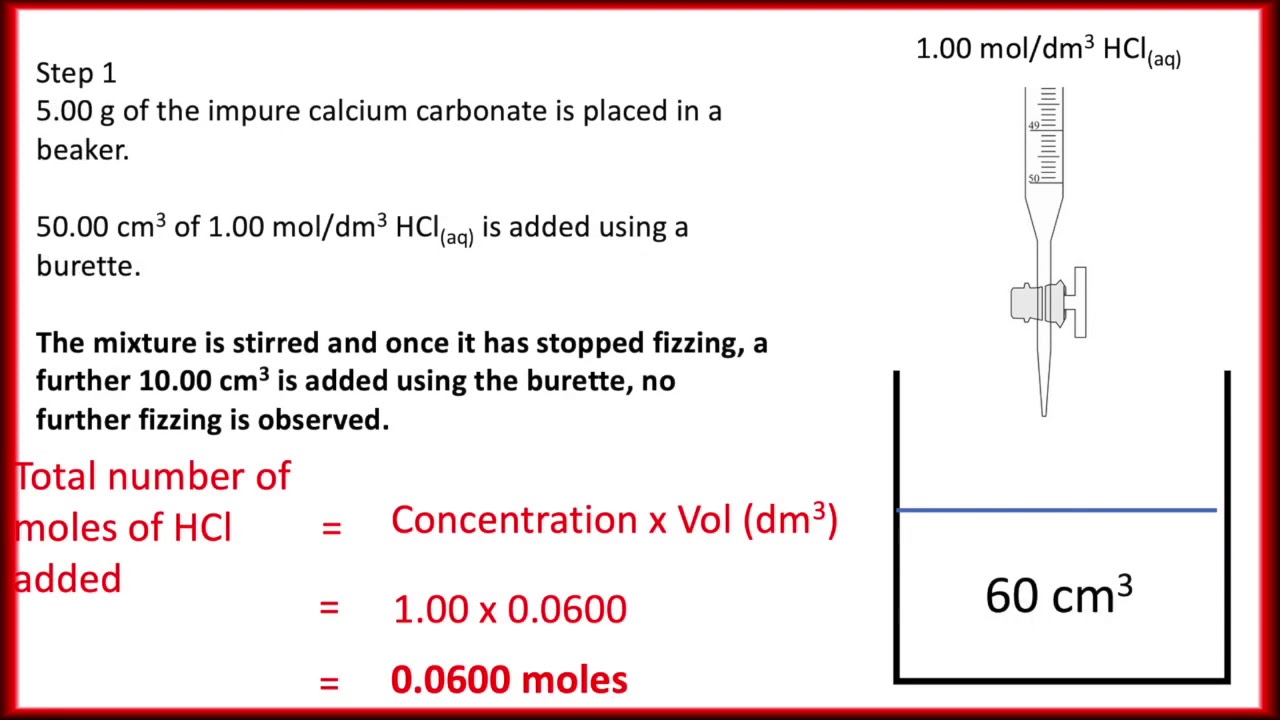
Experiment Report - back titration.docx - Experiment Report Finding the Purity of CaCO3 using Back Titration Introduction In order to calculate the | Course Hero

DOC) OBJECTIVE To determine the calcium carbonate content in eggshell | Sharifah Hana - Academia.edu

Back-Titration F22 - Copy - Back-Titration of a Commercial Antacid Stephanie R. Geggier New York - Studocu

The relationship between the log amount of pure calcium carbonate and... | Download Scientific Diagram

An Extensive Indirect Titration Report Exemplar Regarding the Mass of Calcium Carbonate in Antacids, Received a Final Grading of A+. | Chemistry - Year 12 SACE | Thinkswap






![Lab: Back titration to determine the % CaCO3 in eggshell DATA COLLECTION [IB CHEMISTRY] - YouTube Lab: Back titration to determine the % CaCO3 in eggshell DATA COLLECTION [IB CHEMISTRY] - YouTube](https://i.ytimg.com/vi/fH-RzzDN0Lg/hq720.jpg?sqp=-oaymwEhCK4FEIIDSFryq4qpAxMIARUAAAAAGAElAADIQj0AgKJD&rs=AOn4CLAUKQdGwq7plYa7h8dYUpGwcEOhrA)