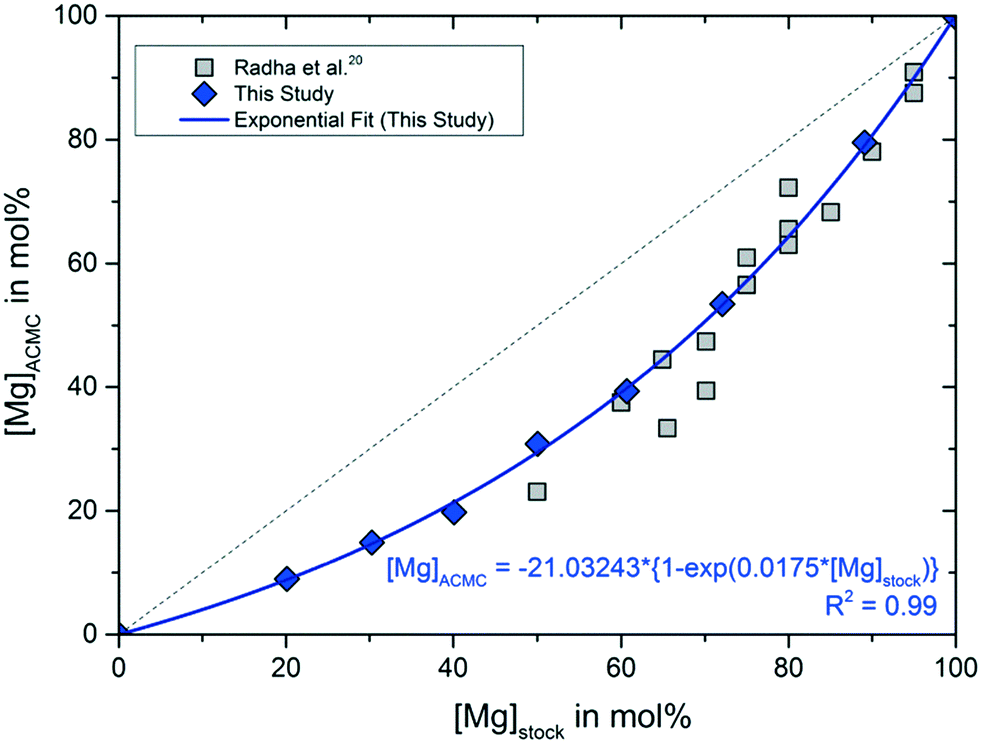
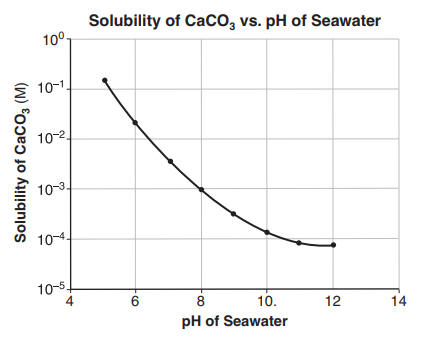
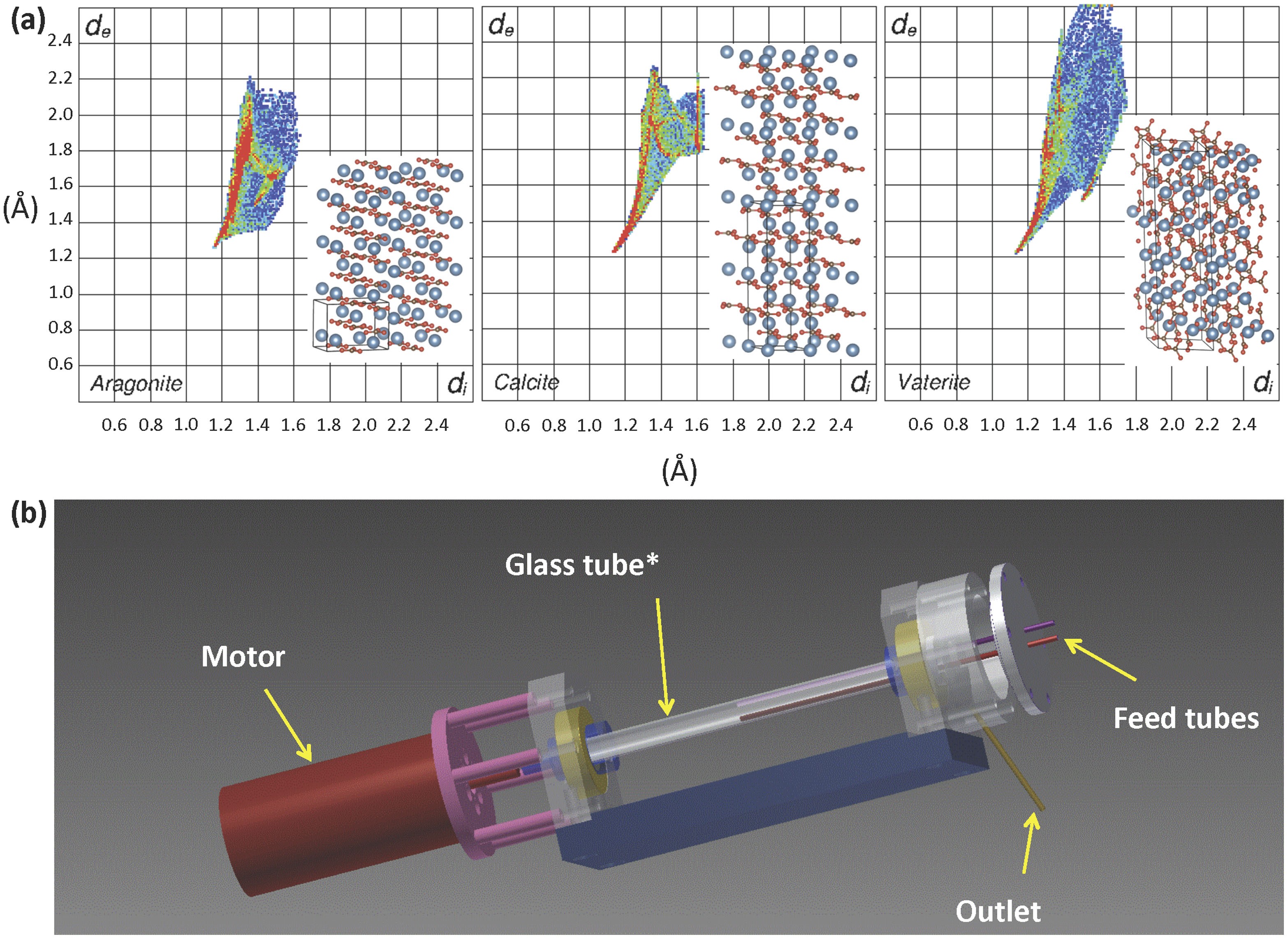
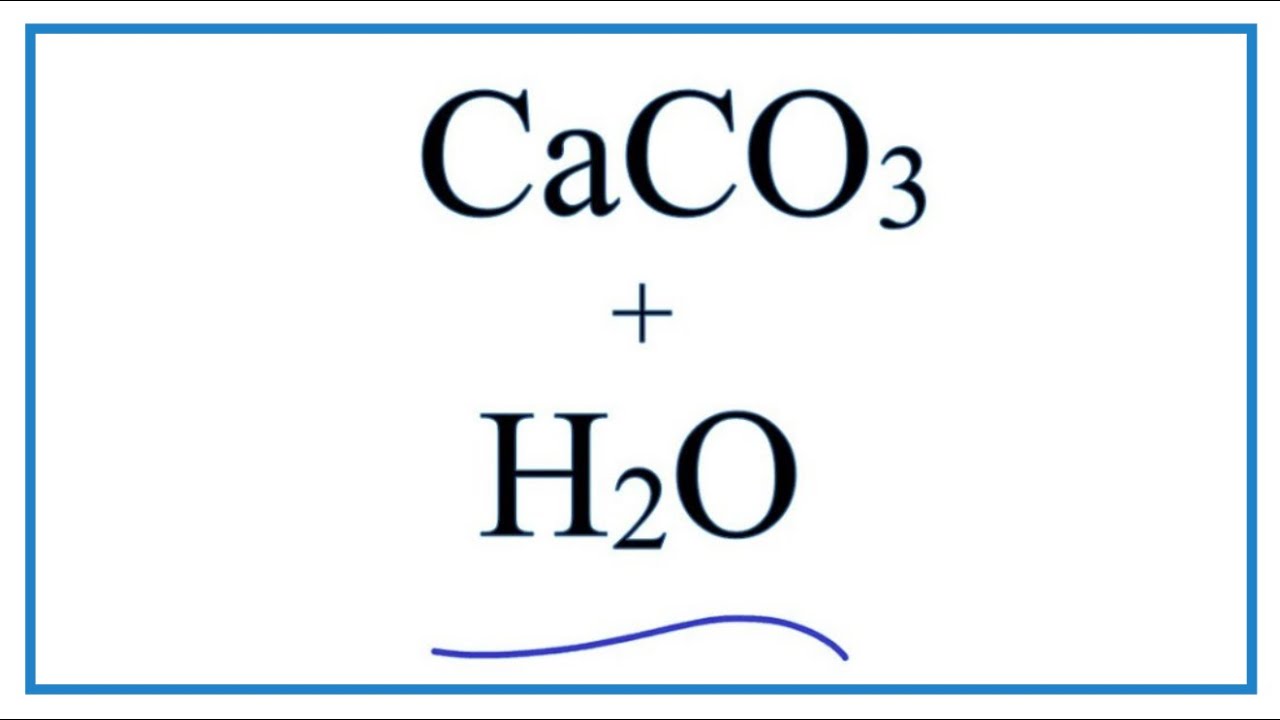
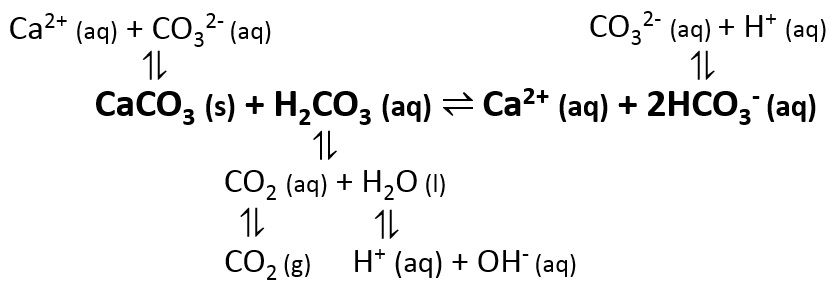Synthesis of calcium carbonate in a pure ethanol and aqueous ethanol solution as the solvent - ScienceDirect

Synthesis of calcium carbonate in a pure ethanol and aqueous ethanol solution as the solvent - ScienceDirect

What happens when aqueous solutions of calcium chloride and of sodium carbonate are mixed? | Socratic

A novel route to prepare the metastable vaterite phase of CaCO3 from CaCl2 ethanol solution and Na2CO3 aqueous solution - ScienceDirect

Uncovering the Role of Bicarbonate in Calcium Carbonate Formation at Near‐Neutral pH - Huang - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Co-precipitation of calcium carbonate and curcumin in an ethanol medium as a novel approach for curcumin dissolution enhancement - ScienceDirect

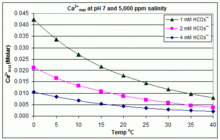
Solubility of Calcium Carbonate in Ammonium Chloride Aqueous Solution at T = (298.15, 323.15, and 348.15) K | Journal of Chemical & Engineering Data
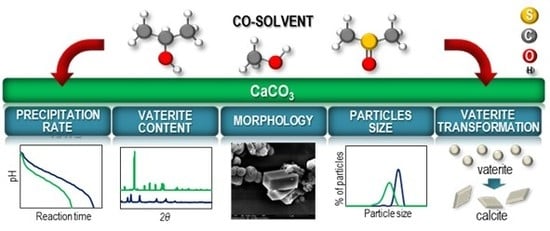
Materials | Free Full-Text | Precipitation and Transformation of Vaterite Calcium Carbonate in the Presence of Some Organic Solvents

Pharmaceutics | Free Full-Text | Ground Calcium Carbonate as a Low Cost and Biosafety Excipient for Solubility and Dissolution Improvement of Praziquantel

Solubility of Calcium Carbonate in Ammonium Chloride Aqueous Solution at T = (298.15, 323.15, and 348.15) K | Journal of Chemical & Engineering Data
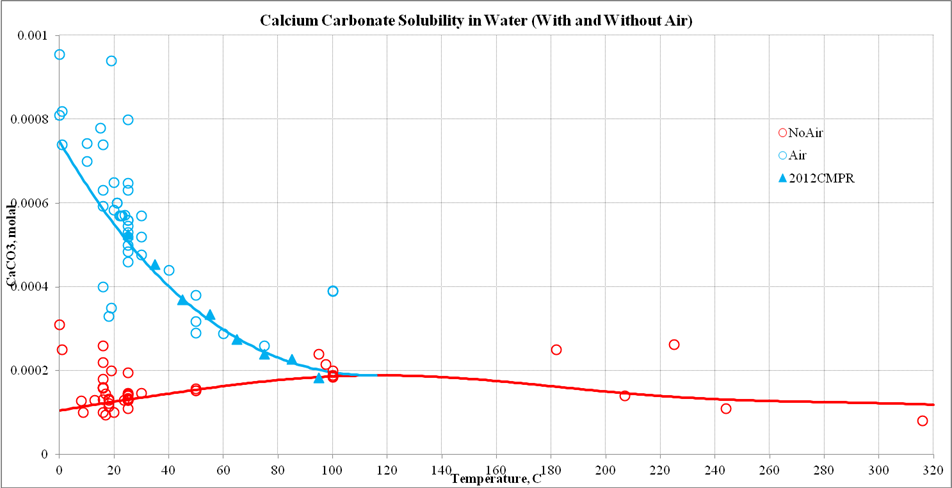
Calcium Carbonate Particle Formation through Precipitation in a Stagnant Bubble and a Bubble Column Reactor

Solubility of Calcium Carbonate in Ammonium Chloride Aqueous Solution at T = (298.15, 323.15, and 348.15) K | Journal of Chemical & Engineering Data
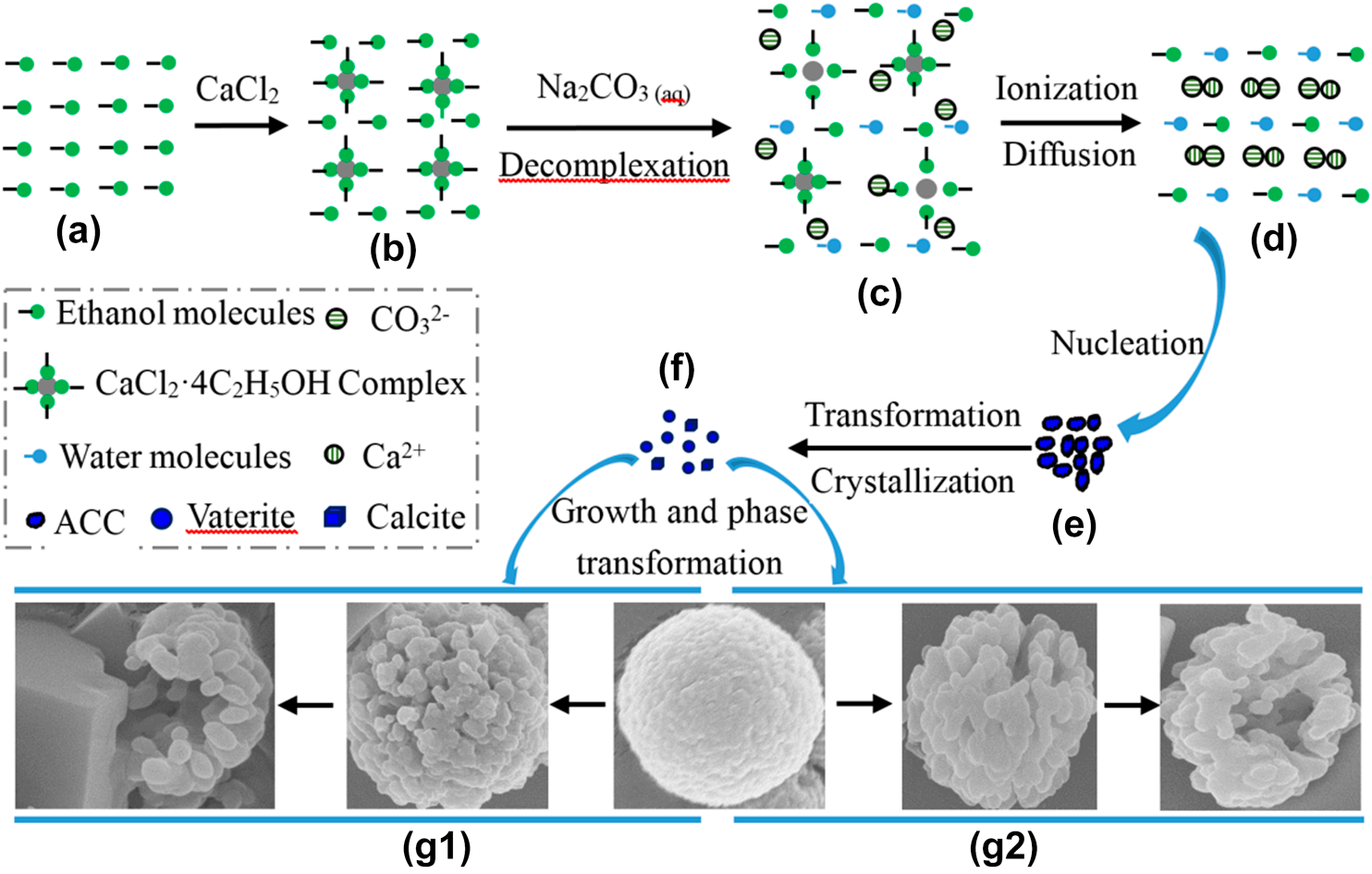
The advantage of alcohol–calcium method on the formation and the stability of vaterite against ethanol–water binary solvent method | Journal of Materials Research | Cambridge Core

Stable Prenucleation Calcium Carbonate Clusters Define Liquid–Liquid Phase Separation - Avaro - 2020 - Angewandte Chemie International Edition - Wiley Online Library
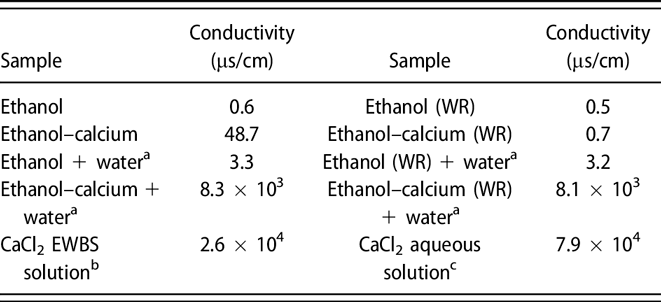
The advantage of alcohol–calcium method on the formation and the stability of vaterite against ethanol–water binary solvent method | Journal of Materials Research | Cambridge Core
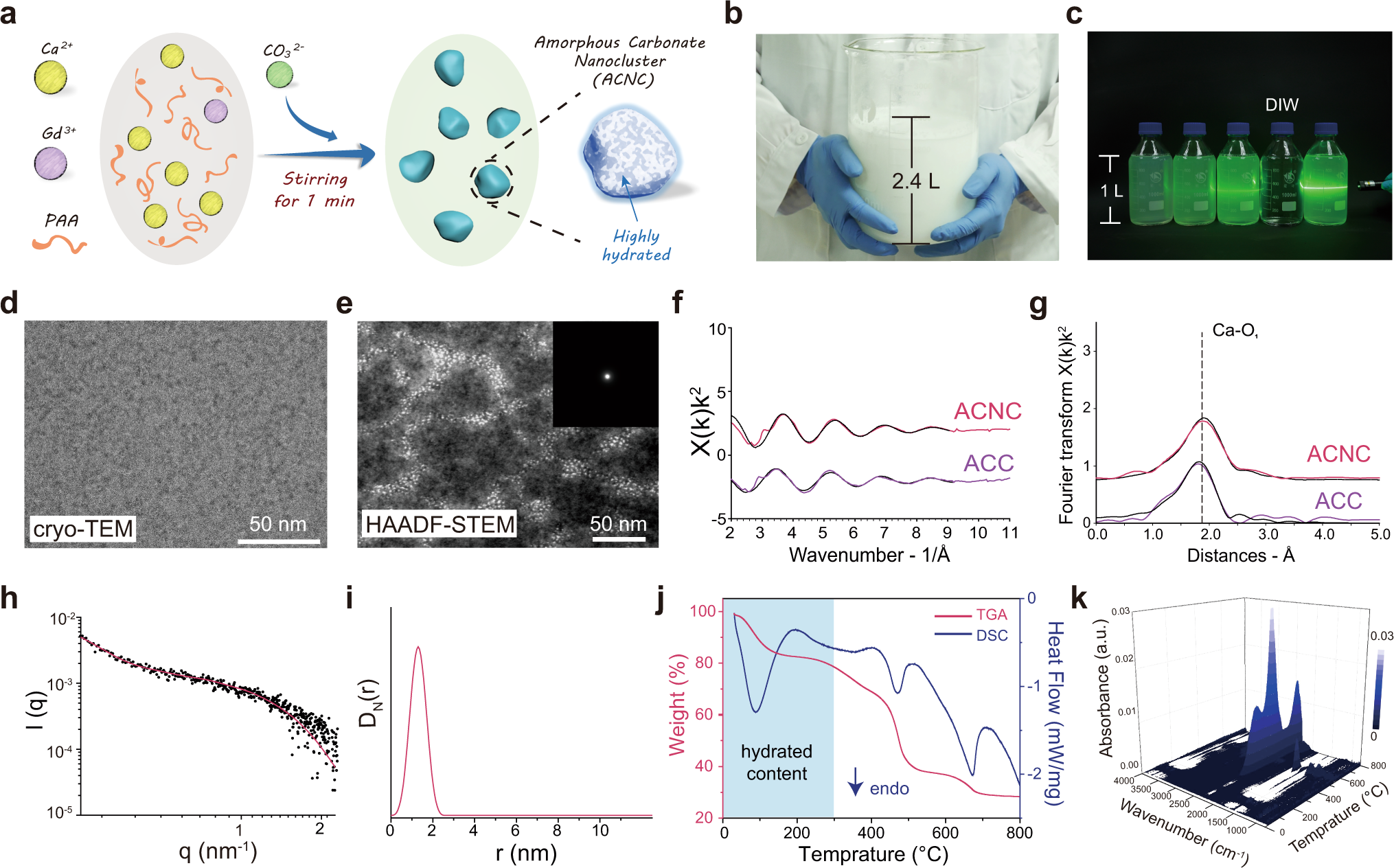
Highly hydrated paramagnetic amorphous calcium carbonate nanoclusters as an MRI contrast agent | Nature Communications
![Adsorption of phenol over bio-based silica/calcium carbonate (CS-SiO2/CaCO3) nanocomposite synthesized from waste eggshells and rice husks [PeerJ] Adsorption of phenol over bio-based silica/calcium carbonate (CS-SiO2/CaCO3) nanocomposite synthesized from waste eggshells and rice husks [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2021/pchem-17/1/fig-1-full.png)
Adsorption of phenol over bio-based silica/calcium carbonate (CS-SiO2/CaCO3) nanocomposite synthesized from waste eggshells and rice husks [PeerJ]