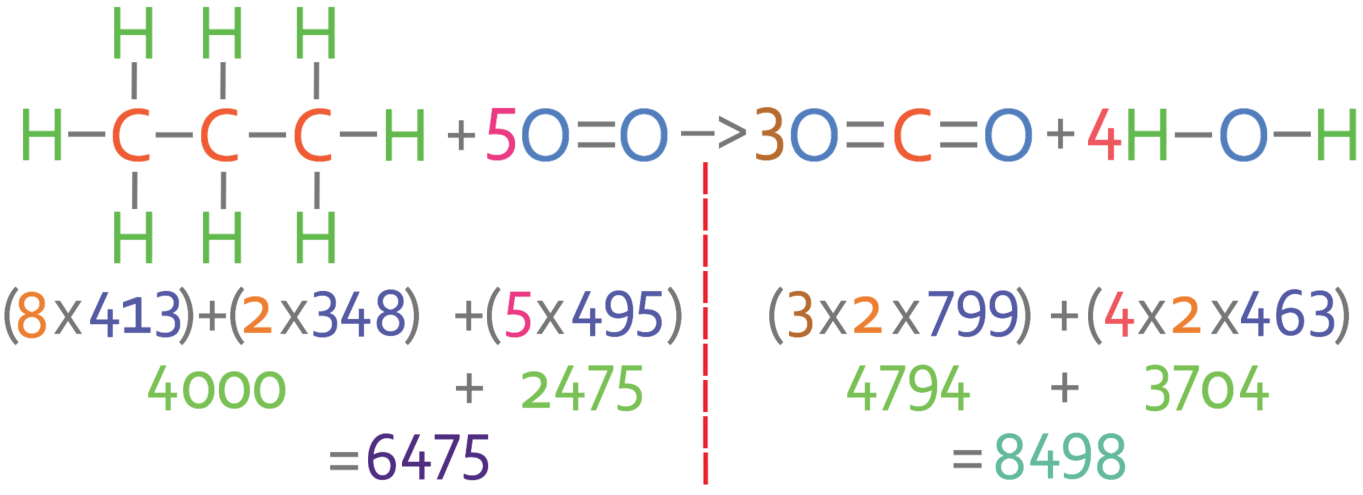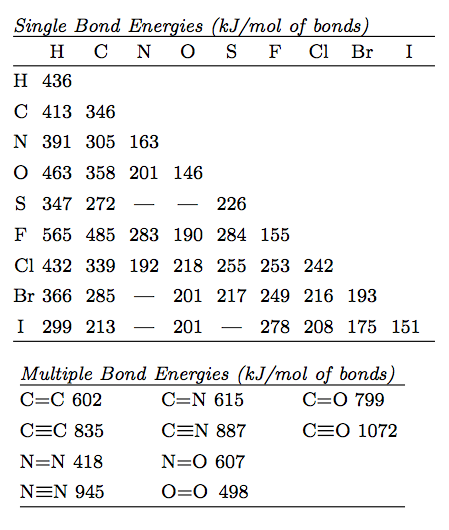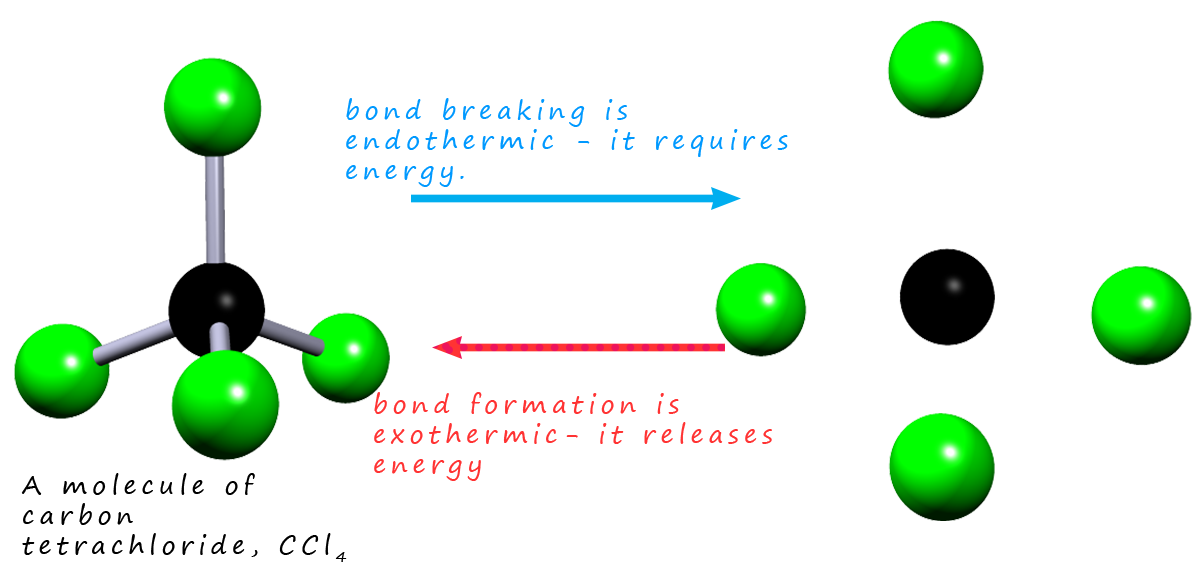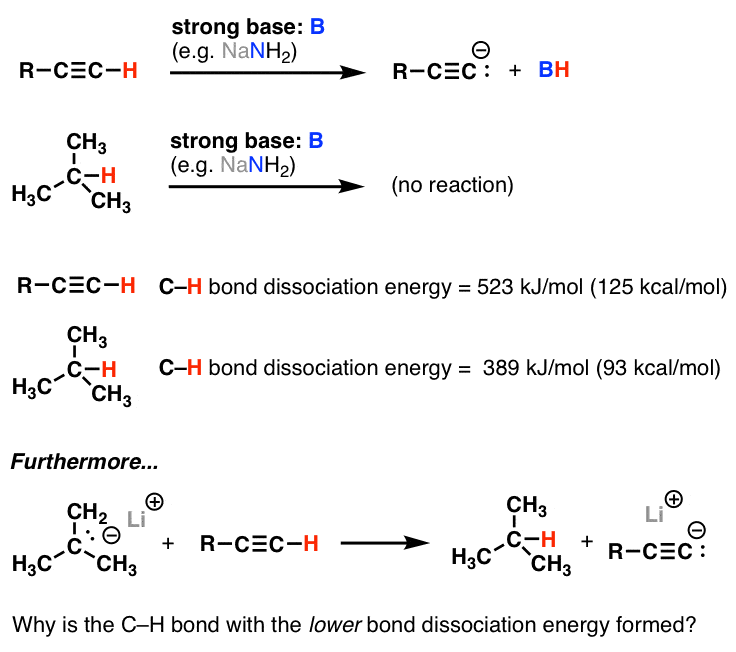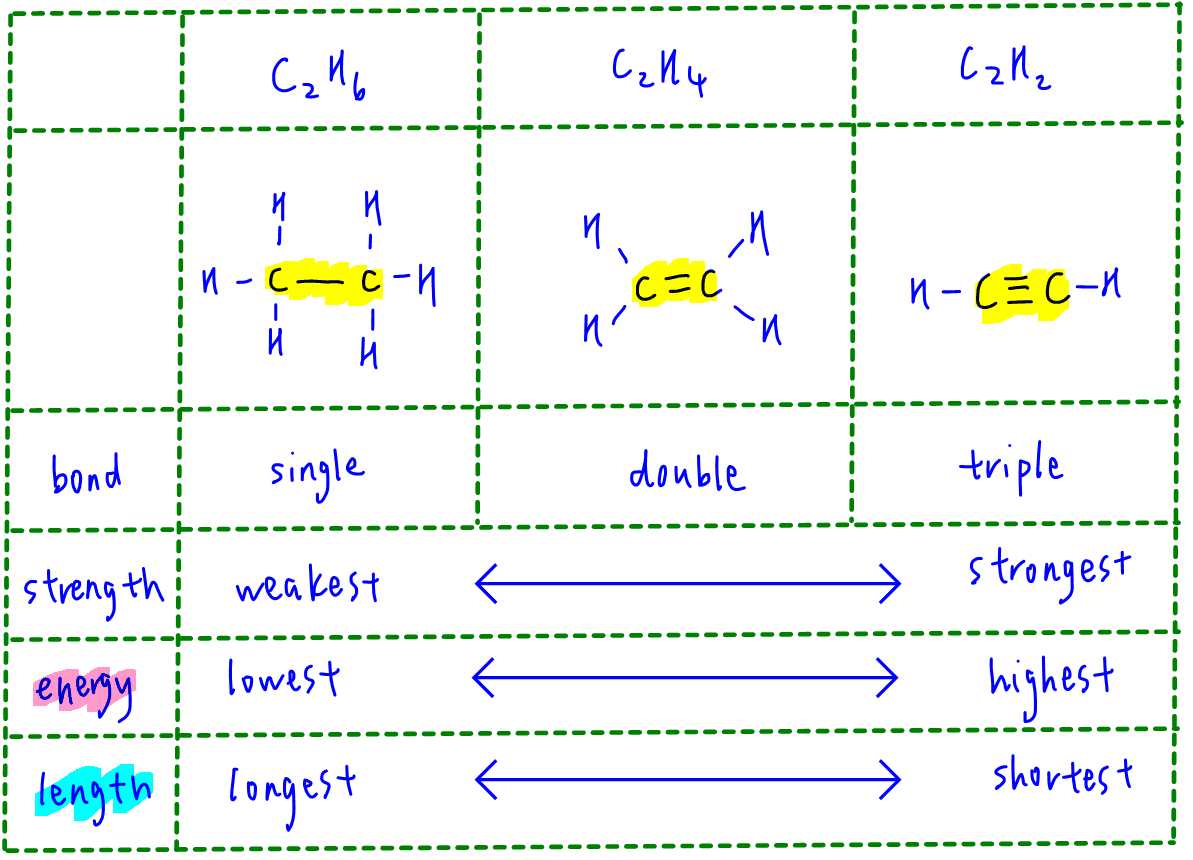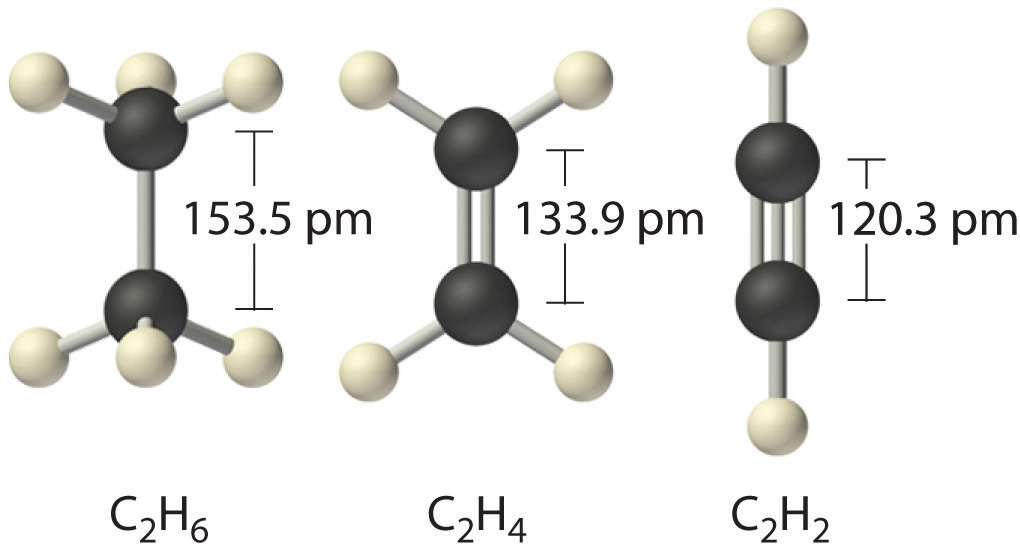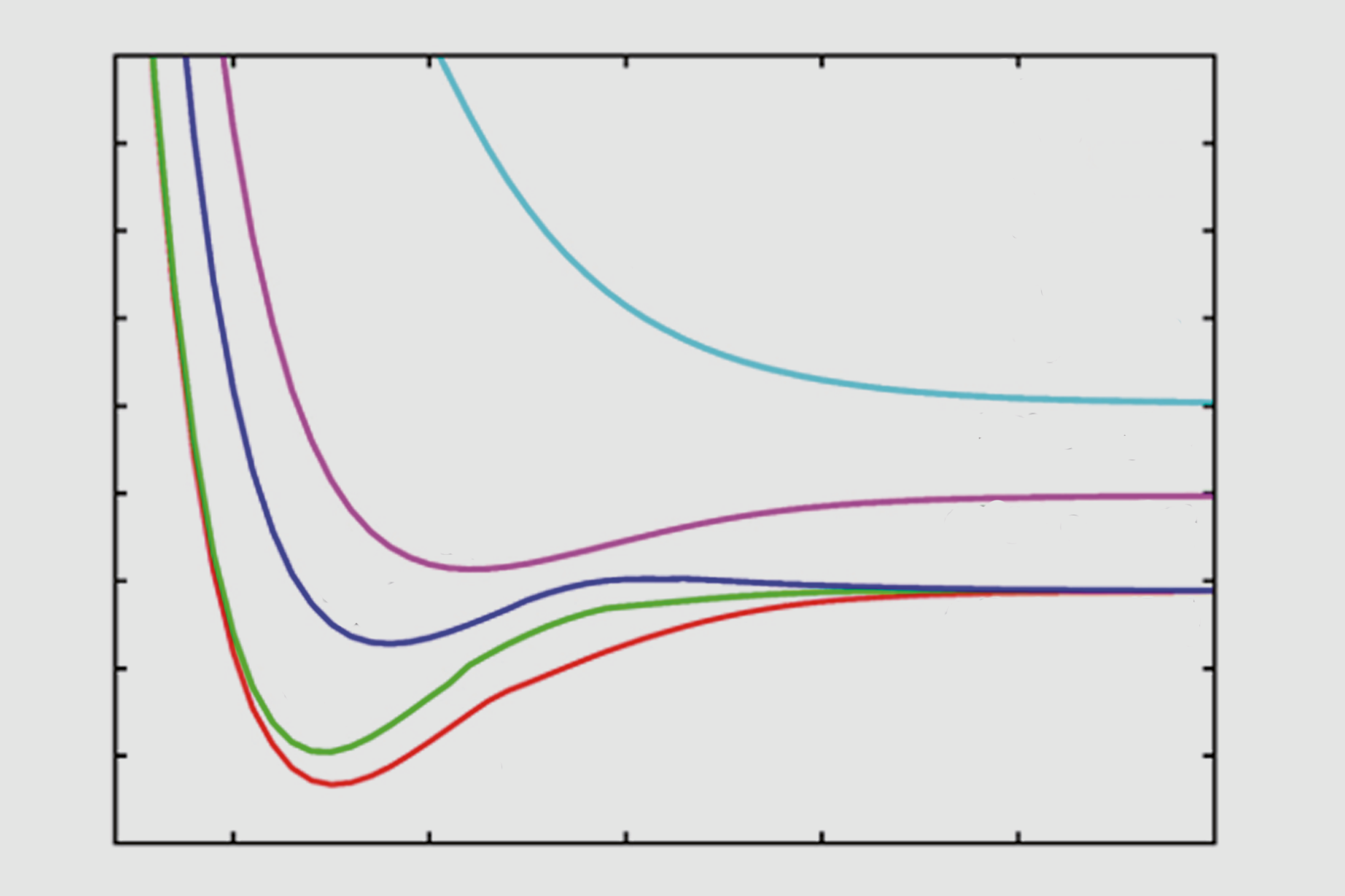
Excited state potential energy curves reignite diatomic carbon's bond order conundrum | Research | Chemistry World

Bonding energy potential and carbon-carbon bond-length distribution.... | Download Scientific Diagram
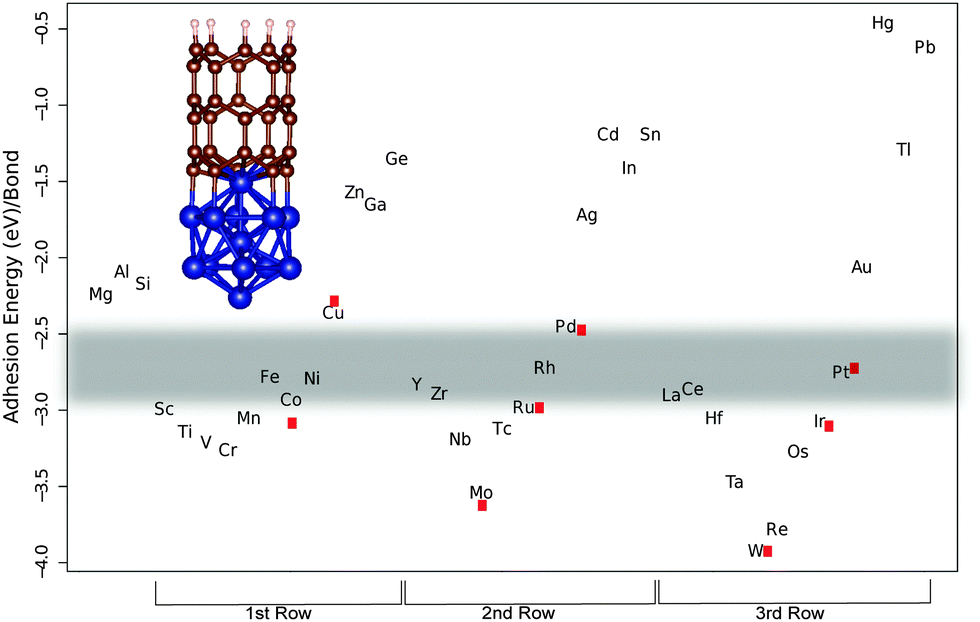
Establishing the most favorable metal–carbon bond strength for carbon nanotube catalysts - Journal of Materials Chemistry C (RSC Publishing) DOI:10.1039/C5TC00143A

physical chemistry - Why is the bond energy for a C=O bond higher in CO2? - Chemistry Stack Exchange
Why are the bonds between Oxygen and Carbon easier to break than the bonds between two Carbon atoms? - Quora
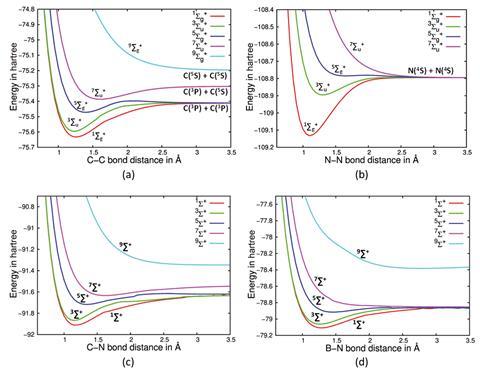
Excited state potential energy curves reignite diatomic carbon's bond order conundrum | Research | Chemistry World