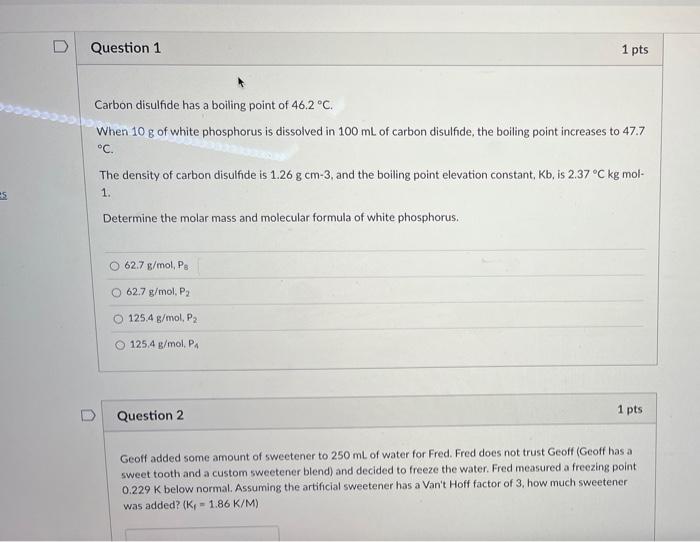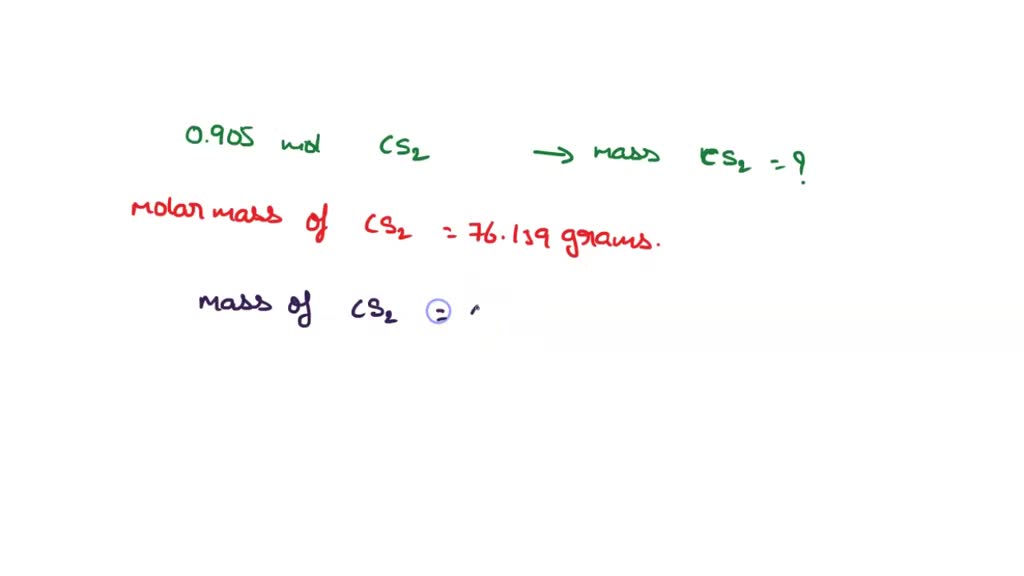
SOLVED: How many grams of carbon disulfide ( CS2 ) are there in 0.905 mol of the compound? 0.905 mol=

Free Online Help: for the above (carbon disulfide if 4.435 g of element are dissolve in 100.0 g of CS2 ) calcualte the atomic weight (or molar mass) of the unknown elements .
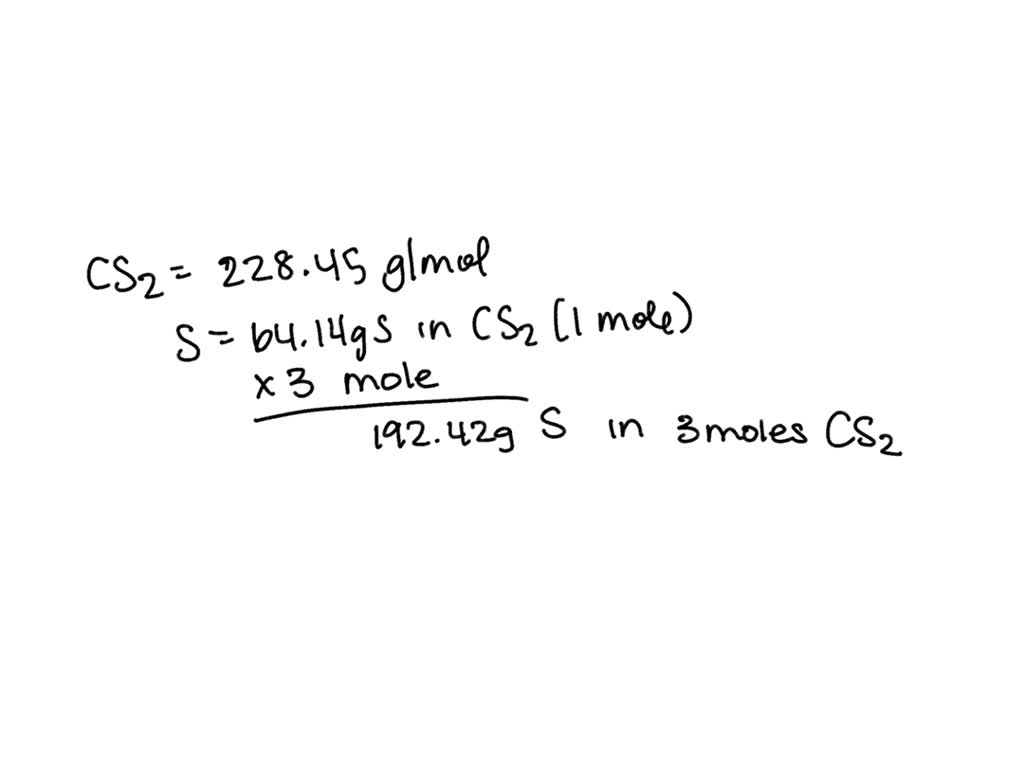
SOLVED: Question What is the mass of sulfur in 3.0 moles of carbon disulfide? 0 96.08 0 64.18 0 32.1 8 Question 8 What is the molar mass of barium hydroxide? 171.3 g/mole 154.3 g/mole 137.3 g/mole 308.6 g/mole 192 8