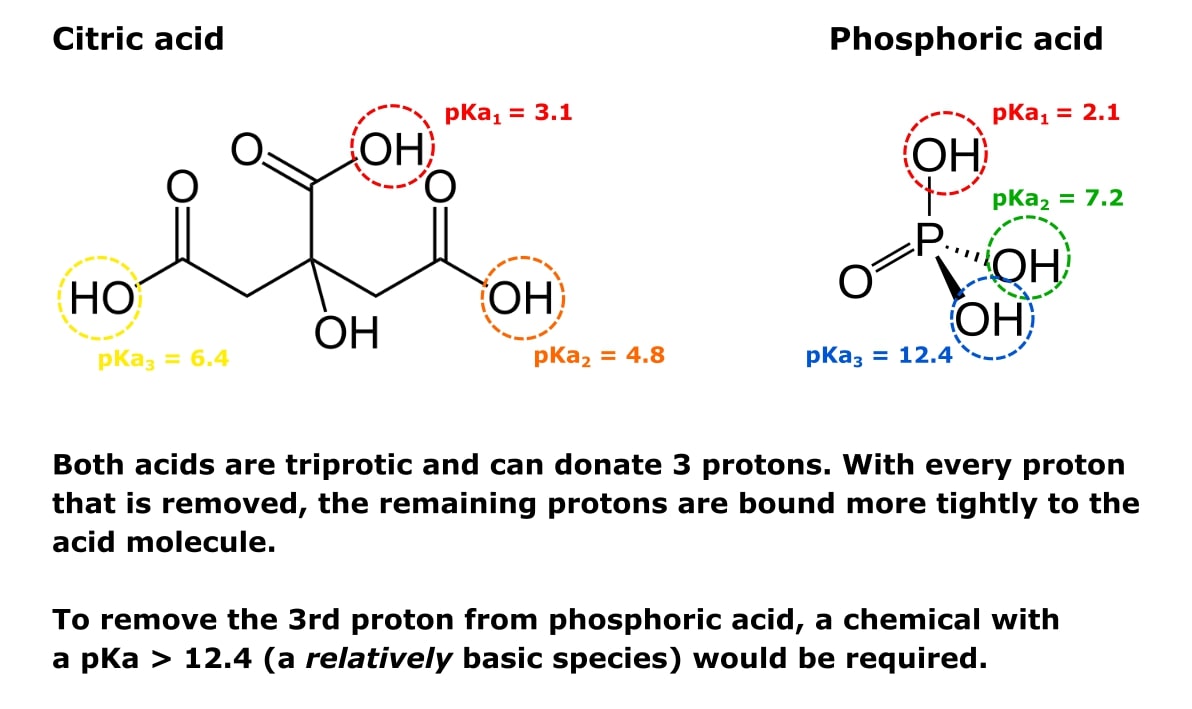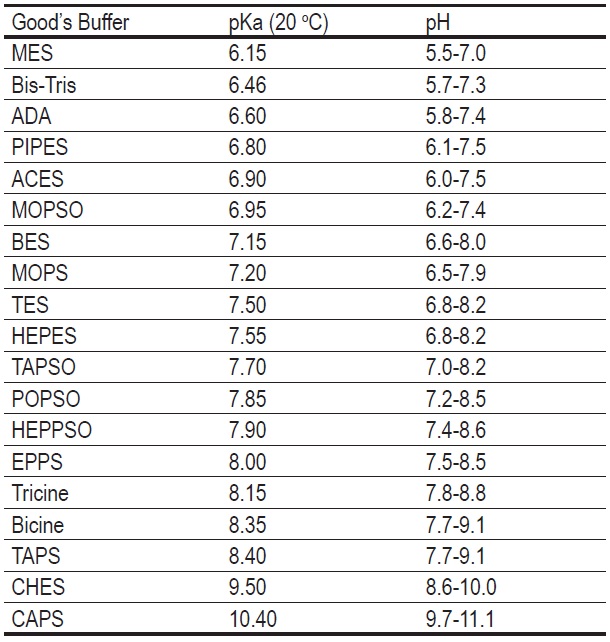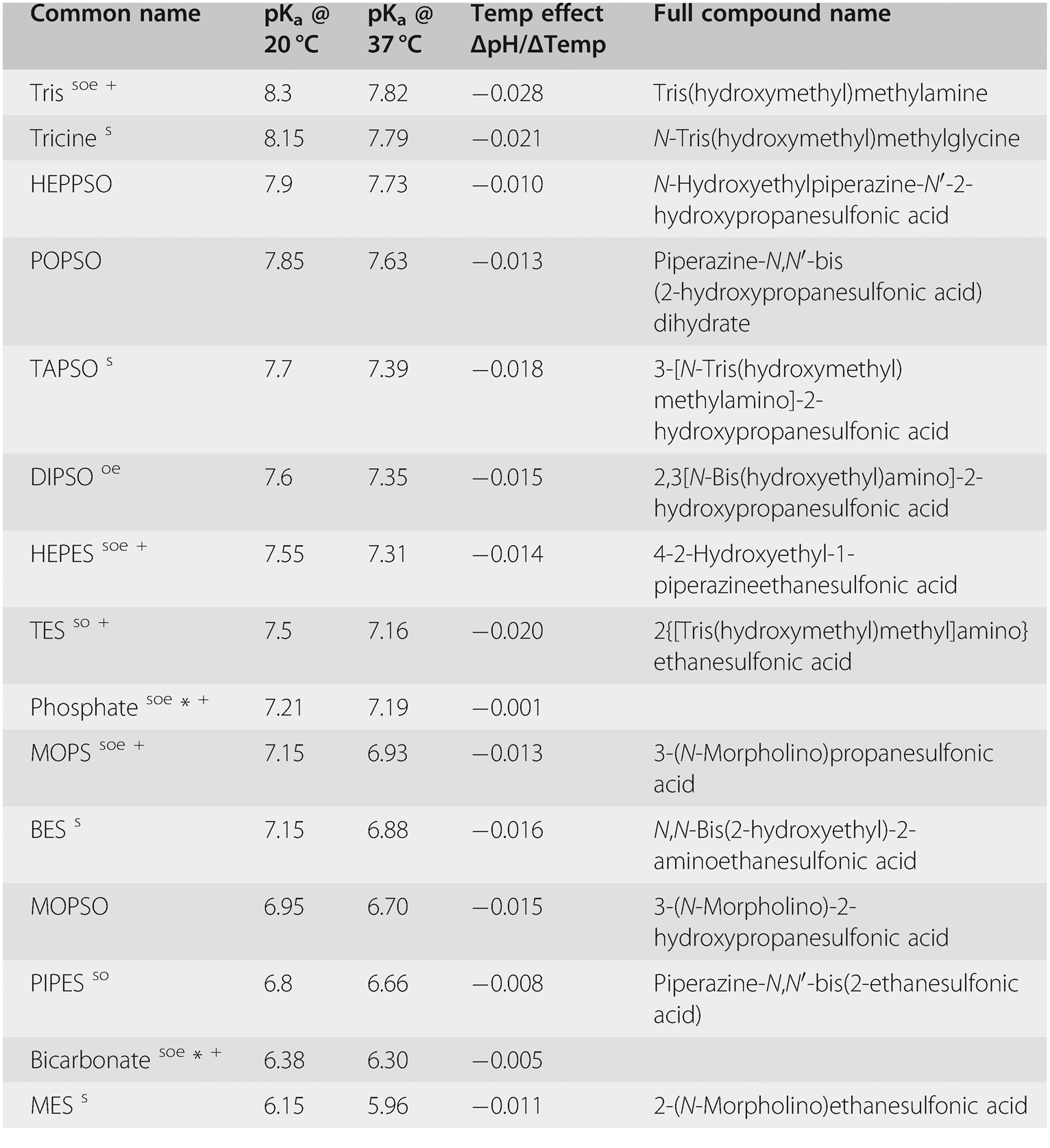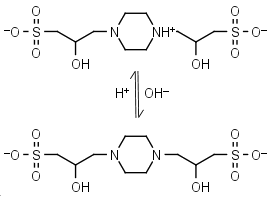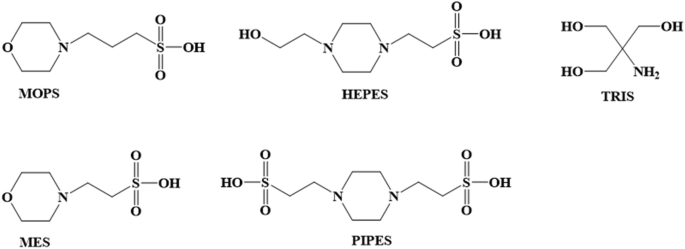
Not just a background: pH buffers do interact with lanthanide ions—a Europium(III) case study | SpringerLink
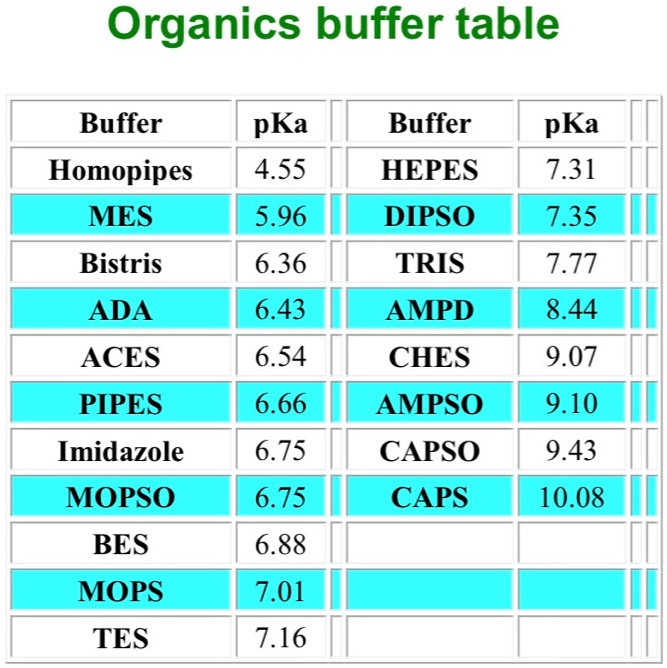
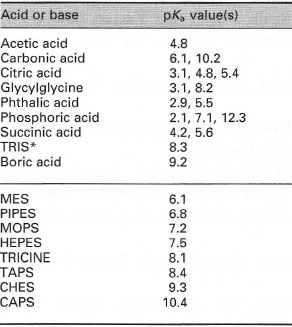
SOLVED: Organics buffer table Buffer Buffer Homopipes MES pKa 4.55 5.96 HEPES pKa 7.31 7.35 DIPSO Bistris 6.36 TRIS AMPD 7.77 ADA 6.43 8.44 ACES PIPES 6.54 CHES AMPSO 9.07 6.66 9.10 9.43 Imidazole 6.75 CAPSO CAPS MOPSO 6.75 10.08 BES 6.88 MOPS TES 7.01 ...

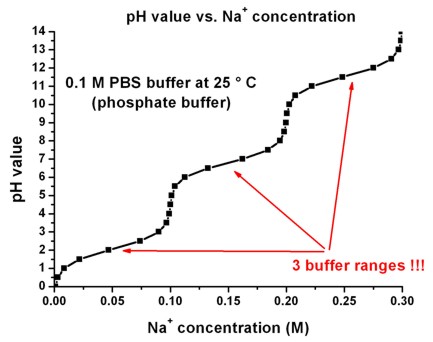
Dissociation steps and pKa values at 25 °C and 37 °C of the buffers... | Download Scientific Diagram
Un)suitability of the use of pH buffers in biological, biochemical and environmental studies and their interaction with metal i

You prepare a buffer solution from 10.0 mL of 0.100 M MOPS (3- morpholinopropane-1-sulfonic acid) and 10.0 mL of 0.077 M NaOH. Next, you add 1.00 mL of 2.05 x 10^-5 M

Dissociation steps and pKa values at 25 °C and 37 °C of the buffers... | Download Scientific Diagram

EP0341793A2 - Buffer solution systems for standardizing ph analyzers and electrolytes - Google Patents

Dissociation steps and pKa values at 25 °C and 37 °C of the buffers... | Download Scientific Diagram


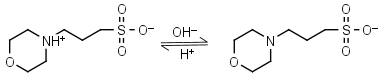
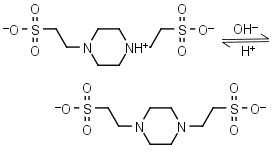
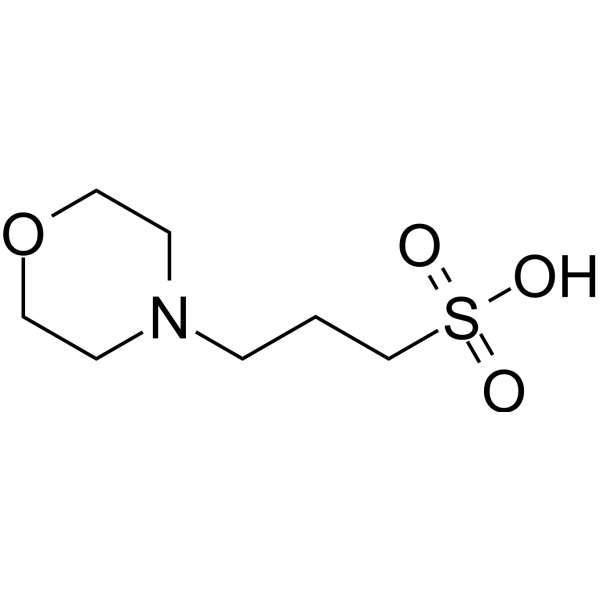

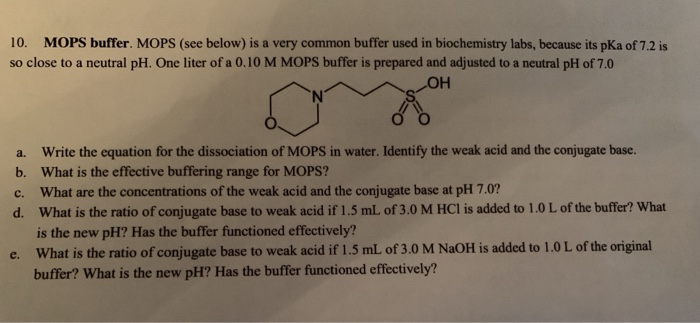
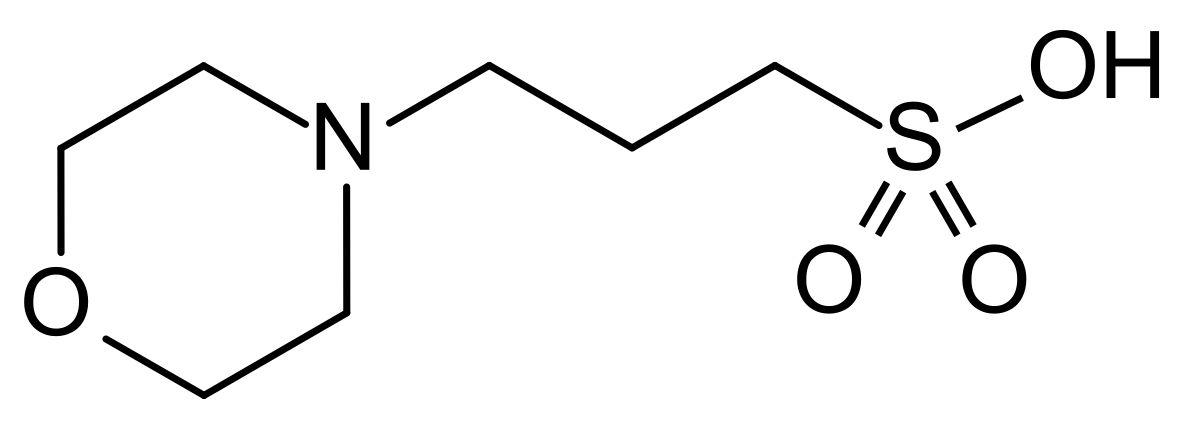
![MOPS Buffer [3-(N-Morpholinopropanesulfonic Acid] - 1132-61-2 - Discovery Fine Chemicals MOPS Buffer [3-(N-Morpholinopropanesulfonic Acid] - 1132-61-2 - Discovery Fine Chemicals](https://discofinechem.com/wp-content/uploads/2015/08/1132-61-2.png)