Molecules | Free Full-Text | Photooxidation of 2-(tert-Butyl)-3-Methyl-2,3,5,6,7,8-Hexahydroquinazolin-4(1H)-one, an Example of Singlet Oxygen ene Reaction
JCM | Free Full-Text | Development of Prodrugs for PDT-Based Combination Therapy Using a Singlet-Oxygen-Sensitive Linker and Quantitative Systems Pharmacology

Comparison of the singlet oxygen ene reactions of cyclic versus acyclic β,γ-unsaturated ketones: an experimental and computational study - ScienceDirect

Formation of 7-carboxyheptyl radical induced by singlet oxygen in the reaction mixture of oleic acid, riboflavin and ferrous ion under the UVA irradiation. - Abstract - Europe PMC

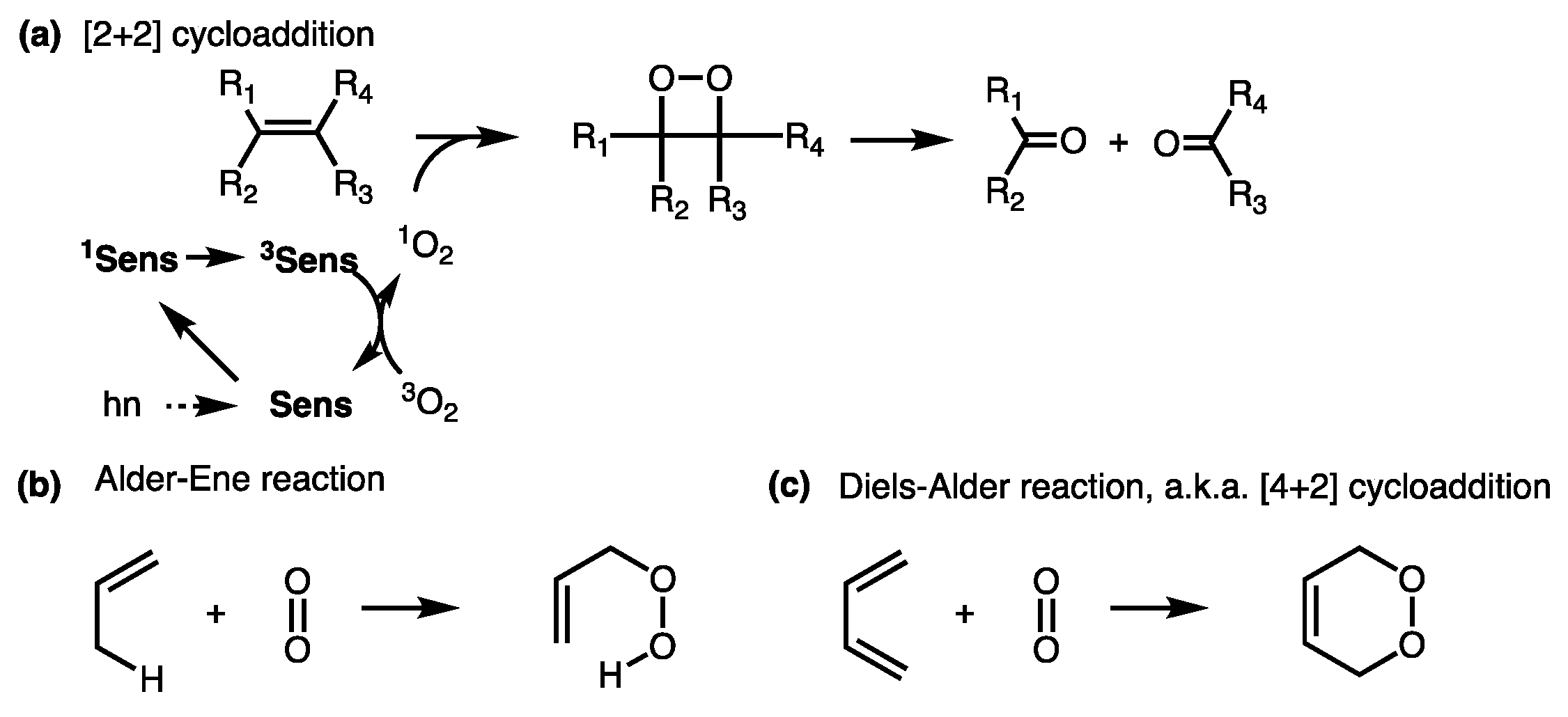
Examples of characteristic reactions of singlet oxygen: (a) A Schenck... | Download Scientific Diagram

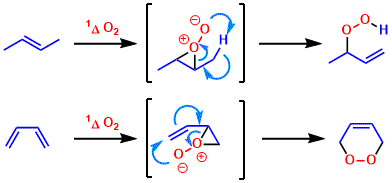
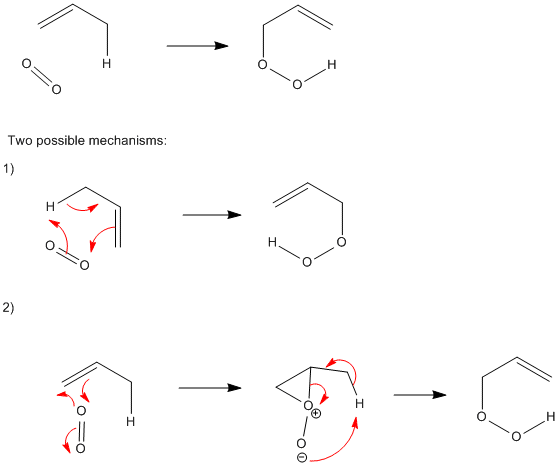
Mechanism of Ene Reactions of Singlet Oxygen. A Two-Step No-Intermediate Mechanism | Journal of the American Chemical Society

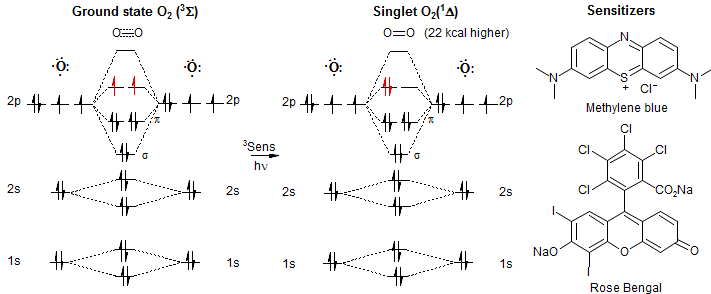
Singlet oxygen and natural substrates: functional polyunsaturated models for the photooxidative degradation of carotenoids

Total Synthesis of Rhodonoids A, B, E, and F, Enabled by Singlet Oxygen Ene Reactions | The Journal of Organic Chemistry

Diels–Alder and ene reactions of singlet oxygen, nitroso compounds and triazolinediones: transition states and mechanisms from contemporary theory - Chemical Communications (RSC Publishing) DOI:10.1039/B111251C
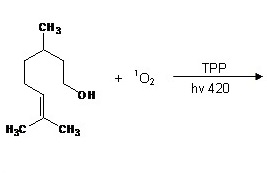
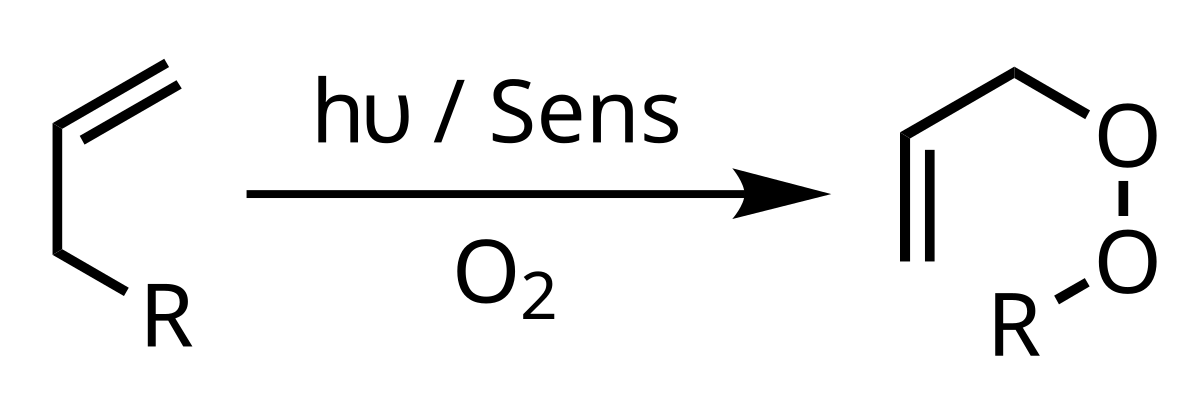
New application note released: Singlet oxygen reactions in flow, an example of the ene reaction - Vapourtec

Mechanism of Ene Reactions of Singlet Oxygen. A Two-Step No-Intermediate Mechanism | Journal of the American Chemical Society

Figure 4 from Mechanism of ene reactions of singlet oxygen. A two-step no-intermediate mechanism. | Semantic Scholar